
This comprehensive reference offers detailed information necessary for understanding and accurately assessing compressor capabilities, as well as for choosing the appropriate designs. Engineers and students will acquire a solid grasp of compression principles, equipment, applications, selection, sizing, installation, and maintenance.
Numerous examples effectively demonstrate essential elements to assist readers in comprehending the practical aspects of compressor technology. The third edition of Compressors: Selection and Sizing has been fully updated to incorporate new API standards.
Enhancements requested by readers include an additional section on diaphragm compressors within the reciprocating compressors chapter, along with a new section addressing rotor dynamics stability in the chapter dedicated to diaphragm compressors. The latest advancements are showcased in the fields of efficiency, 3-D geometry, electronics, CAD, and the utilization of plant computers.
The pivotal chapter on negotiating the acquisition of a compressor now aligns with contemporary industry practices for developing detailed specifications, conducting bid evaluations, performing engineering reviews, and overseeing installation. A significant chapter evaluates the reliability of different compressor types.
Within this book, you will discover all the necessary information to select the appropriate compressor for your specific application. It also encompasses practical insights into compression principles, equipment, applications, selection, sizing, installation, and maintenance. Additionally, new sections on diaphragm compressors and an introduction to rotor dynamics stability are included.

Whether you are a passionate boater aiming to enhance your seamanship skills and secure a discount on boat insurance, or you have ambitions to establish a business operating a charter, sightseeing, or whale-watching vessel, this thoroughly revised and updated Fifth Edition serves as the sole resource necessary for obtaining the captain’s license you desire.
This includes the six-passenger 'Six-Pack,' Master and Mate Inland, Master and Mate Near Coastal, and Sail/Auxiliary Sail Endorsement. The text is easy to read and comprehend, and it also provides tips to assist in memorizing certain information. I highly recommend it if you are pursuing your Captain’s License or simply wish to gain a better understanding of boating practices.
'Get Your Captain’s License' is comprehensive enough to substitute for expensive classroom training, featuring 350 pages of seamanship and navigation tutorials, along with over 1,500 meticulously curated questions and answers from the Coast Guard exams. If you are starting from the ground up, this may not be the most effective method for your learning. However, if you require a detailed refresher on topics you believe you are already familiar with, then this is the most economical way to prepare for the examination.

The MSA Gas Detection Handbook aims to familiarize users with essential terminology and concepts related to gas detection while also functioning as a quick reference guide for information such as specific gas properties, exposure limits, and additional data. This Handbook includes a glossary of vital gas detection terms and abbreviations, along with a concise yet informative overview of fundamental principles in monitoring combustible and toxic gases.
It is abundant in reference data, encompassing physical properties and exposure limits pertinent to the most frequently monitored gases across industrial and various other settings. The author has provided a thorough professional comparison of the most commonly utilized gas detection technologies and has also incorporated a table that outlines the gas hazards typically associated with specific applications in major industries.
Furthermore, a summary of critical information regarding gas detection instrumentation approvals, including classifications for hazardous locations, has been included. MSA’s exclusive Sensor Placement Guide offers detailed insights into important considerations for determining the optimal placement of gas sensors.

Vessels, rather than individuals alone, hold the capacity to alter the course of history. Frequently embodying distinct personalities, ships throughout history have influenced our contemporary lives.
From the formidable fleets of the Persians, Greeks, and Romans to the merchant vessels of the Phoenicians, and from the armadas of Spain and England to solitary ships or small groups of two, three, or four vessels, maritime craft have significantly impacted historical events.
In this intricately detailed book, renowned historian A.A. Hoehling examines these iconic ships: the clippers, the Great Eastern, the USS Monitor, the RMS Lusitania, the ships of Dunkirk, and the USS Arizona.
The author offers vivid portrayals of the ships and their associated figures, vividly recounting the expeditions, battles, triumphs, and setbacks of these extraordinary voyagers. You are encouraged to dedicate some of your time to reading this remarkable book about the most influential ships that truly transformed history, as this is knowledge that everyone should possess.

Sometimes a sedimentary basin is uplifted so that deposition ceases and erosion takes over. An erosion surface is formed; the upper surface of the most recent sediment layer, formerly smooth and horizontal, is modified by running water or other agents.
Later, the region subsides and more sediment accumulates. If the succession of sedimentary layers is thought of as a chronological record, then the buried erosion surface represents a time gap of indeterminate length. Such a gap is called an unconformity.
There are several kinds of unconformity. If the uplift is gentle, so that the rock layers are not tilted or deformed, the gap in the geologic record is termed as disconformities. Although the layers of sediment above are parallel with those below, the shapes of ancient stream channels are often apparent in disconformities. However, the disconformities itself may be parallel with the layers above and below and therefore not readily apparent.
Deposition of sediments on layers that have been deformed and eroded produces an angular unconformity. Sedimentary layers below such nonconformity are not parallel with those above, and the gap in the record is obvious.

This booklet is designed for Licensees, ship operators, shipyards, and marine engineers in general, to illustrate the Unified Technical File created for all MAN B&W engines, which is necessary to comply with the IMO regulation aimed at preventing air pollution.
The Unified Technical File serves as a document that outlines a procedure, primarily based on performance measurements, enabling the operator to confirm adherence to the IMO ‘NOx Technical Code’ to the Flag State Authority or their representative when the engine undergoes checks in service. MAN B&W Diesel has developed this booklet to partly elucidate survey procedures and partly serve as a reference manual for IMO definitions.
The initial section focuses on survey routines, from the testbed to the onboard situation, which is crucial for the operator, detailing the survey process from beginning to end. The subsequent section delves into specific topics and how to manage the most common tasks onboard, such as the spare or reconditioning parts addressed by the regulation. For general emission inquiries or emission control of MAN B&W two-stroke engines, references are made to existing materials published by MAN B&W Diesel.
Many of the initial Technical Files created by engine builders differed due to the varying requirements imposed by different classification societies. This variation occurred because the IMO Annex VI does not provide sufficiently detailed guidance on how to compile the Technical File in practice. MAN B&W Diesel has taken on the responsibility of coordinating efforts to prepare a standardized Technical File, intended for use by both licensees and classification societies. This responsibility also encompasses the necessary procedures for shipowners when subsequent engine adjustments or component changes take place.

Men of the ancient world, throughout their historical timeline, were reluctant to venture far from the sea. It was intricately woven into the fabric of their existence, and among their significant contributions to future generations was their mastery of this exceptionally useful yet perilous means of communication. The ancient mariners of the Mediterranean can be credited with most of the major advancements in shipbuilding and navigation that the western world would recognize until the advent of steam power.
The specifics of this accomplishment—the strategies they developed for rowing war galleys, the rigging they created for merchant vessels, the methods they devised for constructing a hull, and similar topics—constitute a highly technical and specialized field, yet one that is closely linked to the daily lives of ancient people. It is no coincidence that the west's first epic poet chose to narrate the tale of a storm-battered captain, and its first historian and dramatist emphasized a pivotal naval confrontation.
Despite the clear significance of this topic, no scholar adequately addressed it until the very end of the last century. In 1895, Cecil Torr published his work, Ancient Ships, which provided a concise yet commendable overview of the knowledge available at that time regarding the design, construction, and equipment of Greek and Roman vessels.
This publication addressed much of the existing gap, but not all; and although Torr promised further studies on other aspects of shipping, he never completed them. Furthermore, due to the increasing volume of new material that archaeologists were continuously discovering, even his contributions quickly became outdated. Subsequently, after World War II, underwater archaeology emerged and began to explore a source of entirely new evidence...
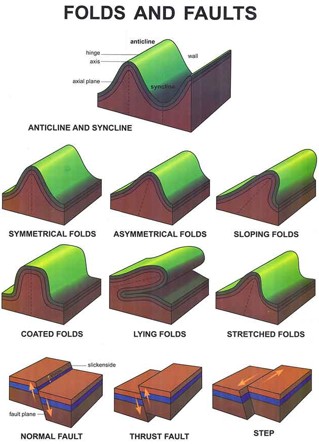
Tectonic plate motion is one of the events that can change the shape and orientation of sedimentary rock layers. Wherever plates converge, the crust is subjected to enormous horizontal forces that can gradually compress it by dozens or even hundreds of miles, wrinkling and folding it like a giant throw rug. The up fold of the crust is an anticline; each down fold is a syncline.
Anticlines and synclines are graphic proof that solid rock can flow like the ice in a glacier. Like most solid materials, rock is slightly plastic under uniform pressure over long periods, it will bend without breaking. However, if stress is applied unevenly or if it exceeds the rock's breaking strength, the rock fractures. A fracture in the crust along which the rocks on opposite sides have shifted relative to each other is termed a fault.
A normal fault is one whose slip plane is at a steep angle with the surface and along which the rock on the upper side has slipped downward in the direction of the dip. In geology, the dip of any surface is the direction in which a marble would roll if placed on it.
A reverse or thrust, fault is one in which the rock on the upper side has been displaced upward along the fault plane. A normal fault allows extension of the crust; it is often caused by forces that stretch the crust. A thrust fault is caused by forces that squeeze the crust together, causing a break where one piece overrides another.
An over-thrust fault is a thrust fault whose slip plane is nearly horizontal its displacement is the result of large horizontal movements of the crust. Along some over-thrust faults, one slab has slipped several miles over the top of another so that a well drilled through the fault would penetrate the same series of rock layers twice.